There are an increasing number of very interesting published studies around genetics / genomics and drug discovery. Just last week, there were a series of articles in Nature on predictors of response to anti-PD1 therapy. (Disclaimer: I work for Merck, which markets an anti-PD1 drug.) In this new blog series, I try and pick at least one paper that highlights some of the key principles of genetics/genomics and drug discovery. This is week #1…hopefully I will be able to do this routinely!
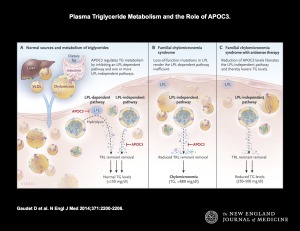
This week, I have selected an article in NEJM by Gaudet et al: Targeting APOC3 in the Familial Chylomicronemia Syndrome. As described in the introduction: “The familial chylomicronemia syndrome is a rare autosomal recessive disease characterized by the buildup in the blood of fat particles called chylomicrons (chylomicronemia), severe hypertriglyceridemia, and the caused by mutations in the gene encoding LPL or, less frequently, by mutations in genes encoding other proteins necessary for LPL function. Patients with this syndrome have plasma triglyceride levels ranging from 10 to 100 times the normal value (1500 to 15,000 mg per deciliter [17 to 170 mmol per liter]), eruptive xanthomas, arthralgias, neurologic symptoms, lipemia retinalis, and hepatosplenomega without pancreatitis, that interfere with normal life and result in frequent hospitalizations.”
Separately, a human genetic study, published in NEJM by Sekar Kathiresan and colleagues (see here), found that loss-of-function (LOF) mutations in APOC3 result in very low levels of plasma triglycerides and a dramatically reduced risk of myocardial infarction. At the time of the publication (June 2014), there was no therapeutic validation that APOC3 was an effective drug target. However, the findings were analogous to the PCSK9 gene, and thus the authors posited that APOC3 is “a bona fide therapeutic target with compelling human genetics validation”. It is worth remembering the size of the human genetic studies required to find these gifts of nature: the genetic association study included >110,000 individuals.
Returning to Gaudet et al…
The therapeutic hypothesis was tested with an antisense inhibitor of APOC3 synthesis, which occurs in the liver. Only three patients were tested, but the results were dramatic within 2 weeks: “Baseline triglyceride levels in the three patients varied (1406, 2083, and 2043 mg per deciliter [15.9, 23.5 and 23.1 mmol per liter]) and fell rapidly during the first 2 weeks in parallel with decreases in APOC3, with triglyceride levels dropping below 500 mg per deciliter in all patients in at least one measurement (Fig. 1B).”
Another interesting aspect of the study was an understanding of human physiology. As stated by the authors in the discussion: “It had been thought that APOC3 modulates triglyceride levels primarily by inhibiting the LPL-dependent clearance pathway, but our findings suggest that APOC3 also strongly regulates the metabolism of triglyceride-rich lipoproteins through LPL-independent pathways.” That is, human genetics linked together with experimental medicine studies that test therapeutic hypotheses in the ideal model organism (humans) can reveal unexpected findings!
Together, the studies by Gaudet et al and Kathiresan and colleagues add to the growing list examples that demonstrate the value of human genetics in identifying targets for drug discovery, especially in genes that harbor LOF mutations that protect from human disease (e.g., PCSK9, NPC1L1, CCR5, SLC30A8).